Stem Cell therapy
Rebuild Joints Using Your Stem Cells
"Rebuild" Joints Using Your Own Cells
Complete Cases
Experience
Rebuild Joints With Stem Cell
If you want want to avoid surgery, and a long painful rehabilitation process, stem cell therapy might be the solution for you. There is some key information to help you decide if stem cell is right for you:
Avoid Costly Surgery
You can avoid expensive surgery and post treatment.
Stem Cell Can Repair Tendons
If you are the right candidate for stem cell, stem cell therapy can actually repair your torn ligament.
Proven Results
Ask about our successful cases of patients who have had stem cell procedures. Read our reviews online.
Caring Staff
Our caring and compassionate staff will walk you through the process, and will help guide you through post treatments.
How Do Stem Cells Work?

Stem Cell Using Your Own Cells
Stem Cell Therapy Can Repair Joints & Torn Ligaments
Joint deterioration is usually a progressive degenerative disease. That means it does not get better on its own and the joint cartilage will gradually wear away. Damaged joints cause restricted, or altered movement, which often leads to other problems such as weight gain, digestive problems or foot problems.
The Stem Cell Process
In order to achieve the highest stem cell counts possible, we use multiple activation methods to isolate the highest number of stem cells. Many stem cell clinics use a single activation method, while others neglect this step altogether.
We include PRP treatment in our stem cell procedure, to further facilitate the stem cells in “doing their job”.
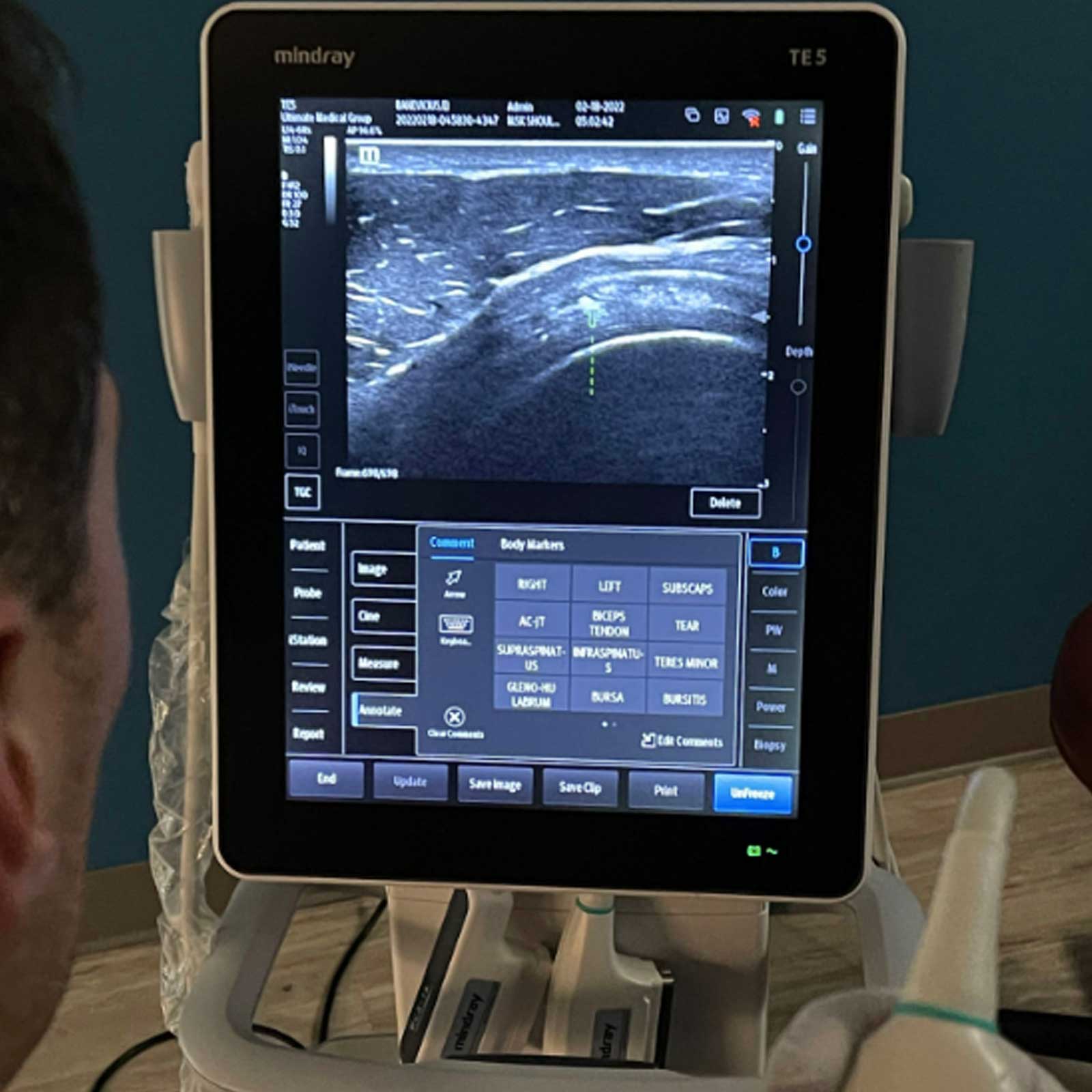
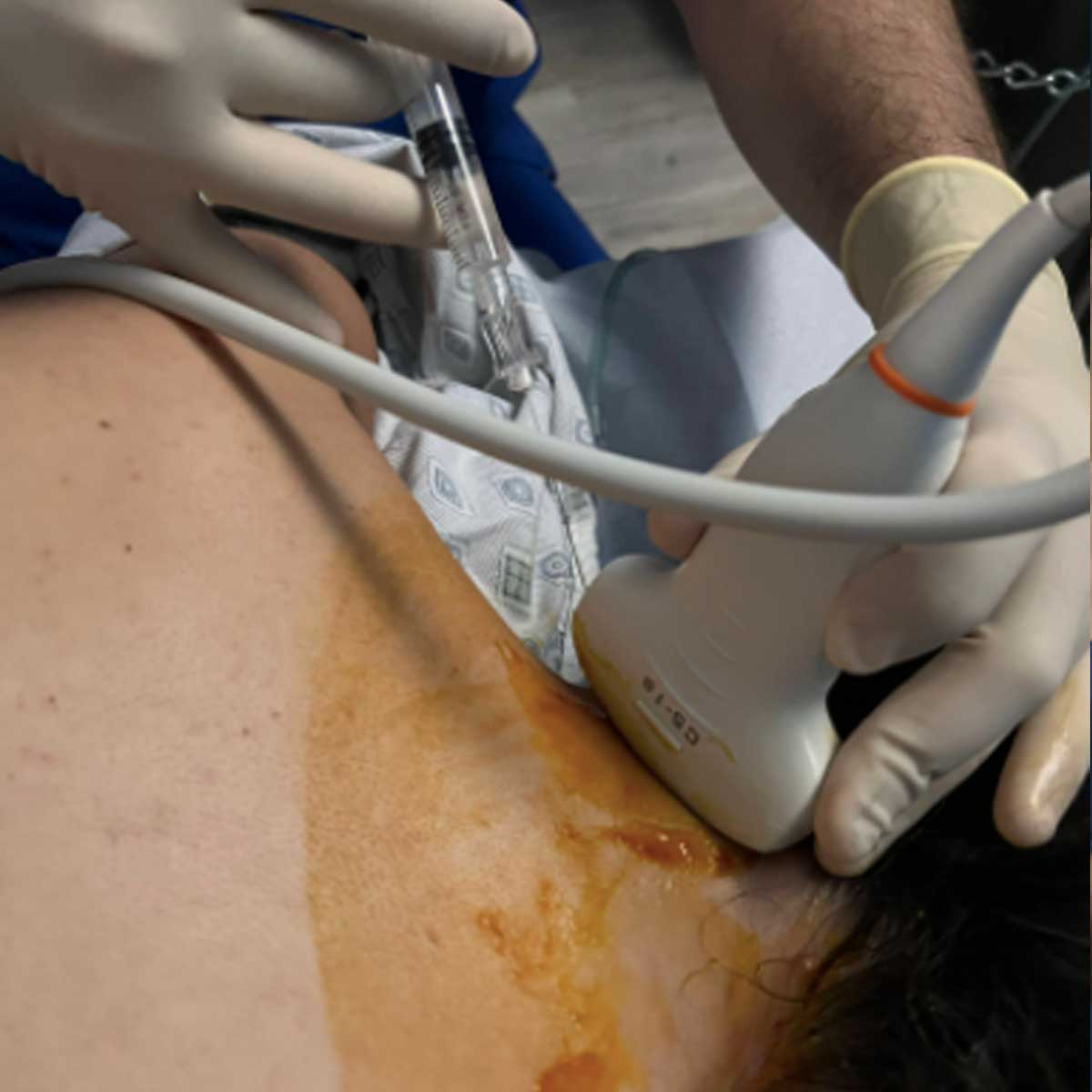
Using high definition ultrasound imaging, we precisely inject the high concentration of cells into the injured area.
Key Benefits
01. Certified Ultrasound Specialist
Our medical director is highly experienced and is a certified diagnostic musculoskeletal ultrasound specialist.
02. Bone Marrow Derived
We use bone marrow stem cell aspiration, stem count, and viability technology that has minimal downtime.
03. No Donor/Substituted Stem Cells
Frozen, over the counter stem cells have inherent risks associated with any purchased product. There is no substitute for using your own stem cells to facilitate healing.
Activating Stem Cells Post-Treatment
A critical component of our success with stem cell procedures is following up with specialized therapy, specific to the area of injury.
Your stem cells need an optimum environment to do their job. Your injury was likely a result of a structural imbalance, long-term repetitive use or, less frequently, an acute injury.
In order to “activate” the stem cells, our entire team has developed highly effective post-procedure protocols to facilitate the healing process.
Physical therapy is recommended following your procedure. If your injury was a result of a structural imbalance, it’s important to correct the problem, allowing the stem cells to work as efficiently as possible.
Stem Cell Therapy FAQs
FDA Compliance & Patient Safety
Ultimate Medical Group is committed to providing safe, ethical, and compliant care in accordance with applicable FDA guidelines and medical standards. We prioritize transparency and patient education so individuals can make informed decisions about non-surgical treatments designed to support healing, function, and mobility. The FAQs below address common questions about FDA oversight, treatment safety, and our approach to responsible care.
Yes. The processing kits used are provided by Emcyte Corporation and all of these devices are 501K certified by the FDA. These devices produce some of the highest concentrations of PRP, BMAC & A2M on the market today.
Yes. The processing kit used to produce Micro Fat Stem Cell Injections is provided by Jointech Labs. The device is 501K certified by the FDA.
No. The FDA categorizes “Stem Cell Therapies” as “Biologics”, and Umbilical Cord Stem Cells are regulated by the Public Health Service Act and the Federal Food, Drug, and Cosmetic Act. At the present time, the only FDA use of Stem Cell Therapies derived from umbilical cord blood are for blood related conditions such as Leukemia and Lymphoma. Any use of Umbilical Cord Blood for Orthopedic and Neurologic conditions is not approved by the FDA.
No. Exosomes are regulated and need approval for use in human conditions. AT the present time, there is no FDA approved use of Exosomes for Orthopedic and Neurologic conditions.
NO. These products are considered Biologics under section 351 of (PHS ACT), and drugs under sections 201 of (FD&C ACT). Any Clinic offering these products must file an IND application (Investigational New Drug) with the FDA. At the present time, there are no FDA approved products for Orthopedic and Neurologic conditions. Any clinic offering these products without full FDA approval and regulation is in violation.
Watch Our Video Library On Stem Cell
How We Developed Stem Cell Therapy
0:16
0:16
0:16
Our Expert Team

Dr. Perry Cammisa

Dr. Kory Begy

Dr. Nicholas Angelopoulos






